 On the nature of allosteric transitions: a plausible model. The influence of pressure on the rates of biological reactions. Application of a theory of enzyme specificity to protein synthesis. Binding energy, specificity, and enzymic catalysis: the circe effect. Structural mechanisms for domain movements in proteins. The influence of configuration on enzyme activity. Induced fit, conformational selection and independent dynamic segments: an extended view of binding events. Conservation of mus-ms enzyme motions in the apo- and substrate-mimicked state. 
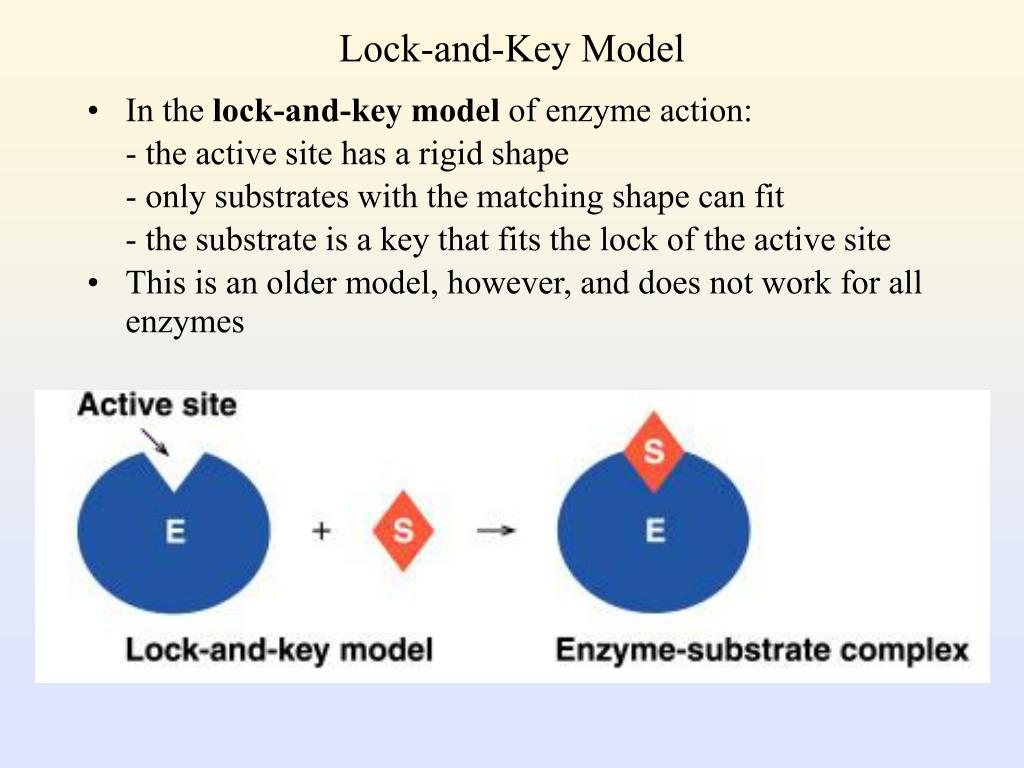
The reaction is then catalysed and an Enzyme-Product Complex forms.Beach H, Cole R, Gill ML, Loria JP. When a substrate molecule collides with an enzyme, if its composition is specifically correct, the shape of the enzyme’s Active Site will change so that the substrate fits into it and an Enzyme-Substrate Complex can form. It states that the shape of Active Sites are not exactly Complementary, but change shape in the presence of a specific substrate to become Complementary. According to this model, it is possible for an enzyme to catalyse a reverse reaction.Ī more recent model, which is backed up by evidence ,and is widely accepted as describing the way enzymes work, is the Induced-Fit Hypothesis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
